Living in northern BC, I have a lot of acquaintances that use wood stoves to heat their home. I really don’t want to say I feel superior to them, but gas is cleaner, better for the environment, and more convenient. Unless something interrupts the gas supply, I have a reliable source of heat. While they’re chopping wood like peons, I can just turn on the thermostat and be toasty warm. Actually, I DO want to say I’m superior to them. BWAHAHAHA! HAHAHA…
…HAHA…
..ha..
…Oh, well. To celebrate my impending hypothermia, I’m having a special all-methane blog post!
Natural gas is essentially methane; the gas is fairly abundant in northern BC. And before anyone starts looking for them, I’m not making any of the obvious jokes in this post.
First, some photos I took of the explosion. They’re not as good as the ones on the news reports. To be honest, I’m a little disappointed; by the time I got to Prince George and took these photos, over an hour had passed since the explosion. On the other hand, I’m still impressed; I was over 20 km away, and could still see the flames. There were no injuries or damaged property(other than the pipeline itself), otherwise I wouldn’t be as cheerful. If it had happened back in August, this post would have a different tone.

The second photo was taken about half an hour later, near the University of Northern BC(probably about 18 km away). The glow was still present, but getting dimmer quickly. The daylight could be described the same way, so it still made for a good photo.

When I took the above photos on Tuesday, the twin to this pipeline was also shut down as a precaution. After it was examined and found to still be safe, it was reactivated on Thursday. The second pipeline is much smaller than the ‘explodey’ one, so we’re still trying to cut back on using natural gas. From what I understand, we’ll still have enough gas, but the farther south the pipeline travels, the more the gas will be used up. Without conserving it, Southern BC might start having shortages. In other words they’d better stay on Northern BC’s good side, otherwise we might all take long, hot baths… that sounded more intimidating in my head.
The pipeline carries the gas as far south as Washington… well, right now it doesn’t, which is kind of the problem. It’s reducing the natural gas to some petroleum refineries in Washington, which will likely drive up gasoline prices. It sounds like it will be a couple weeks before the pipeline is repaired, so we’ll see what the effects are.
Propane is much more efficient(with about 2.5x the energy from the same volume), but is not produced here. If propane was in the pipeline, I would have had some GREAT photos… Likely ones I would have taken while driving as far away as possible.
No photos outside the visible spectrum of the fire; I didn’t have my other equipment with me, and didn’t have time to go back and pick it up. Instead, some photos from under more controlled circumstances.
Last summer, I was taking photos of a flame from a gas stove…look; I get bored sometimes.

The stove is on the ‘high’ setting. This was during fire season- the sunlight is muted and brownish, and it was somewhat dim. As I was taking a picture of a flame, that wasn’t a problem- the dimmer, the better.
Unlike wood fires, the gas from the stove mostly burns blue. Every now and again, there is a little flicker of orange; this seems to be from dust igniting or an impurity. Generally, though, the flame is very even. It doesn’t jump up or flicker, so it is easy to do a comparison between different photos.



The flames look similar in the red, green, and blue channels. It is slightly brighter in the blue channel; but the surrounding kitchen is darker, so it might be partially an illusion(not entirely, though- the pixels in any given location are demonstrably brighter in the blue channel). The red channel is slightly dimmer than green and much dimmer than blue- the burner itself is most visible in the red channel.
A side by side comparison isn’t the best way to see the difference in the channels, however. When the differences are so subtle, I find that an animation works better.

So, the flame is clearly brighter the shorter the wavelength. Does that trend continue?
At the full spectrum level, things start to change.

The flames are noticeably higher. They have a pinkish hue- as I adjust the white balance for each wavelength, this is a false colour photo. The hue is invisible to the naked eye, and is a response to something outside the visible spectrum. As for what the higher flames indicate, it is hard to tell from this picture alone; I’d assume that it was from infrared light, as my camera is less sensitive to ultraviolet. That was my working hypothesis, though; looking through different filters would help resolve the issue.

With the short band pass filter, ultraviolet, violet, and some blue light passes into the camera. I’ve mentioned before that I cannot get a proper white balance with this filter; this is as close as I could get. It looks cool without any balancing, so here’s that picture.

The flames are bright, but not very high. The exposure was longer, too. The high flames don’t seem to be coming from the UV end of the range.

With my Schott UG11 filter, the flames are bit higher, but as it allows both ultraviolet and infrared light, it still doesn’t narrow anything down. I’d need to block one or the other.
So, my ultraviolet photo…. doesn’t exist. The dual band photo exposure took over 2 seconds, and from experience, ultraviolet takes 20-30 times as long(ie 1/15 second vs 4 second exposures are normal). Everything is working against ultraviolet light; the haze from the smoke cuts it down, the glass windows block more, and my camera itself isn’t very sensitive to it. There is likely no way to get a photo with a shutter speed of less than a minute.
Instead, infrared.

The photo with my Zomei 680 nm filter clears up the question of where the ‘pink’ flames are from- the fire is obviously higher in infrared. A photo taken with the Zomei 760 nm filter…

…shows the flames are even higher in a longer wavelentgh. This trend continues with my Zomei 950 nm filter.

The flames are much higher, reaching to the top of the jars in the background. Despite the fact that the flames from the stove look bluish, longer wavelengths show more activity. Red just seems to be less present.
But, does a longer wavelength photo mean the flames are higher? My Canon Rebel T3i full spectrum camera(I took all the above photos with that camera) can’t see much beyond 1000 nm. My low resolution Seek Reveal thermal camera can, however.

That is much higher; it looks like a wood fire. his photo was taken at the same location as the other photos; THere’s no zoom function, but if I take the photo farther away…

The thermal infrared light seems to reflect off the table. It isn’t as warm as the fire(obviously, as building coded frown on countertops being above the boiling point of water). I couldn’t get a good reading of the temperature in the heart of the flames- the highest I recorded was 236° C, but obviously the flame is hotter than that. If I tried to get closer, the temperature would exceed the limit for the camera. Instead, I’m focusing on the flame as a whole.
This was taken with the ‘white filter’ setting; the lighter it is, the hotter.With the ‘cool filter’ setting, the flame looks like this:


One thing that I noticed with the flame in the thermal range is that the top moves and flickers with the air currents much more than it does in the visible range.
Finally, just to show off a few other ‘filters’ for the thermal camera, here is the gas stove with the ‘iron’ filter settings
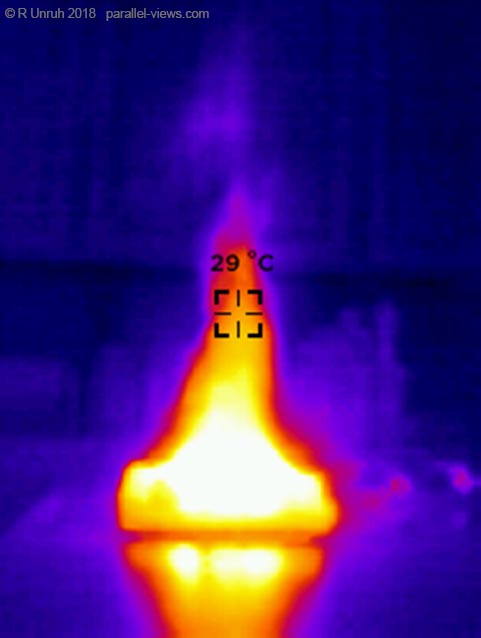
… and with the ‘Tyrian’ settings

But enough about natural gas. It’s time to sit back, scan the news, see if Vancouver has done anything to annoy me, and then look for any reason to turn the thermostat WAY up.
